The world first learnt the benefits of nuclear radiation with the work of Marie Curie more than 120 years ago. Today we know all that we need to know scientifically, but unfortunately people remain unfamiliar with it and so are easily but unnecessarily spooked. In particular they are concerned about nuclear waste.
What is high-level nuclear waste made of?
A mixture of
- unused fuel, “fissile” material usually uranium-235 or plutonium-239. Not particularly radioactive, it can be handled with gloves.
- other “fissionable” material, such as uranium-238 or thorium-232. Even less radioactive.
- fission products, a vast spectrum of species including caesium-137, strontium-90, and iodine-131. Highly radioactive with short half lives of up to 30 years.
- plutonium and other actinides like americium-241. These are produced in a reactor and are fissionable. Many have long half lives, many thousand years.
Is it dangerous?
It is nasty, but in nearly 70 years of operating nuclear power stations nobody has lost their life from coping with nuclear waste - a record as good as the waste product from any competing industrial process. Treated sensibly it is safe:
Is there much of it?
The waste is a product of the fuel and, because the fuel is so energy-rich, the quantity is minute in both cases. The energy density of nuclear fuel is a million times that of coal. So, whereas the energy needs of one person for life can be provided by a thousand tons of coal, just one Kg of pure uranium could do the same. That means the choice of waste is between a million tons of carbon dioxide and a Kg of nuclear waste if fully used. (This figure is subject to a few ifs-and-buts, factors of 1/10 to ten, though insufficient to change the conclusion.)
Is it valuable?
When waste is removed from a reactor, the fuel is only about 1-2% used - such “waste” would better be called “slightly used fuel". As such it can deliver 50 times more energy and is therefore very valuable. However, the fission products need to be removed first otherwise they poison further energy production.
Also valuable are the unique by-products in nuclear waste, essential for use in clinical medicine, normal smoke detectors, and more.
Can it be used to make a nuclear weapon?
To make plutonium-239 for a nuclear weapon the nuclear fuel in a reactor must be removed after a very short time. Any longer and the used fuel becomes “poisoned” by plutonium-240 from which it cannot be separated. This constant replacement of fuel is not compatible with the operation of a reactor designed to provide a steady source of electricity. A nuclear explosion cannot be ignited with fuel contaminated by plutonium-240. So nuclear waste is not a source of nuclear proliferation.
Can it be used to make a dirty bomb?
A dirty bomb assumes that the “dirty” radioactivity of used fuel is spread out by the blast of a conventional high explosive and that the radiological contamination will be seen as immensely harmful. The high explosive itself may be damaging but the radiological consequences would be very small compared with the fear excited among those recipients who are scared at the mention of radiation.
When fear kills: the case of nuclear energy. A lecture given at the William Dunn School of Pathology, Oxford University (See also pdf version of slides).
Is nuclear waste likely to pollute the environment?
No. Since it is composed of heavy elements (strontium is one of the lightest, eight times heavier than carbon), most of the waste is solid with a high boiling point. As such it is already captured and does not leak into the environment. (The carbon dioxide released into the environment by burning fossil fuel is a million times greater in quantity and there is no technology to re-capture it to scale.)
The best demonstration that nuclear waste stays put is given by the story of the Oklo Reactor . This makes an easy but fascinating read and tells how nuclear waste buried in the ground two billion years ago has not move substantially since - seventy times older than the age of the dinosaurs!
Radioactivity released inside the Earth finds its way to the surface as radium dissolved in water. These spa waters and their radon are much sought after for their therapeutic value. Today this is true in traditional Japanese hot springs and Baden in Germany - and was in the baths of ancient Rome, too.
What should be done with high level nuclear waste?
When, and whether, nuclear waste should be reprocessed for its unused fuel is an economic question. If the cost of fresh fuel goes up, partially used fuel should be recycled. Anyway, immediately after removal from the reactor it is physically hot. Storing it under water for ten years keeps it cool and safely absorbs its radiation from the external environment.
How long does it remain radioactive?
Within minutes of a reactor shutting down, the production of new nuclear energy ceases. What continues is heat production by the radioactive “embers”. Initially this is 7% of the “on” power of the reactor but it falls progressively. After a year it is less than 0.1%. The red curve below shows how its activity drops below that of natural uranium after a million years - a time that excites popular worry.
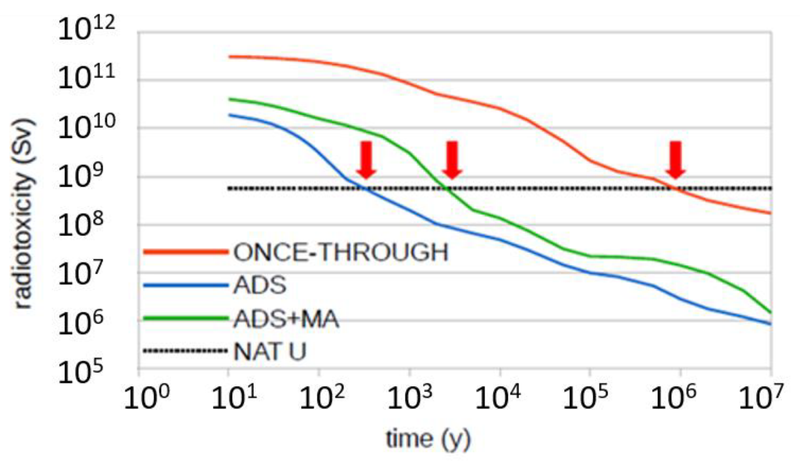
But this concern is misplaced. The waste products of fossil fuel combustion contain significant quantities of cadmium, arsenic and other chemical poisons that never decay at all. Without public comment these are buried, out of sight and unnoticed. In the case of nuclear waste the rapidly decaying fission fragments described by the blue curve become insignificant after a few hundred years. (The contribution of the minor actinides (MA) are included in the green curve, but these are fissionable and can be recycled.)
After cooling under water spent fuel can be air cooled, stored in dry casks, recycled and vitrified. To put the risk in perspective, consider that the radiation level in the nuclear waste storage hall at Sellafield is so low that anyone would have to stay there for a million hours to receive the same dose that a radiotherapy patient receives to healthy tissue in a single day.
The use of long-term deep geological burial is unnecessary and a waste of resources. The point is that after the heat declines there is no agency to drive a runaway process like there is in a chemical fire or a runaway battery “fire” The only uncontrollable process is the public reaction to the idea of nuclear waste - and this is best extinguished through education and public information. This is important and requires attention.
Whoever suggested that it was a problem? And why did they do that?
After the bombing of Hiroshima and Nagasaki opinion at all levels became terrified of nuclear weapons. More precisely, they did not trust the authorities to refrain from using them.
During the Cold War concern grew as arsenals increased worldwide. Public demonstrations, vocally supported by Einstein and other Nobel Prize winners, provided a fertile environment for theories supercharged with caution. Although there was no reliable evidence to support it at the time, the assumption that nuclear radiation is irreversibly dangerous to life was accepted and with it that nuclear waste is dangerous.
Today this view finds no objection from the fossil fuel industry, nor from the industry that has grown up dedicated to providing protection against radiation. In fact, it is well established today that low exposures are beneficial to health, but most experts with an interest are loath to accept this conclusion, yet.
A final thought
Every atom around us, except hydrogen, is nuclear waste. Every carbon, nitrogen and oxygen atom was made in nuclear explosions before the Solar System was formed 4.5 billion years - similar explosions are seen elsewhere in the Universe today. The residual radioactivity still persists as the uranium, thorium and radioactive potassium-40 found in our environment. These atoms have half lives of many billions of years and are still decaying. In fact their decay heats the inside of the Earth, causing volcanic eruptions and earthquakes. In March 2011 this process driven by “natural” nuclear waste caused 20,000 deaths and was accepted stoically by Japanese society; it also triggered the Fukushima accident in which “manmade” nuclear waste caused no deaths - but a panic that spread worldwide.
