Science is the great antidote to the poison of enthusiasm and superstition. - Adam Smith
Summary. The nations of the world plan to stop burning carbon fuels, but have not fixed on the replacement. For social and economic confidence, they need to share a proper picture of the options. The science is simply explained and not in doubt, though widely misunderstood. Energy sources belong to three distinct groups – “renewables”, chemical and nuclear. Since human life began it has adopted each of these in turn. In the past the initial disruption has been more than off-set by the rise in human values that followed. Now, to complete the final step we answer those who would look backwards to the age of “renewables”. Instead, the world should look forwards to a heavy dependence on nuclear energy with a confidence, informed by natural science and openly shared in society.
Energy in quantity
Accounting for energy is like keeping a financial balance sheet — in fact more so, since natural science holds energy to a gold standard. It dictates that energy is conserved and cannot be created or destroyed, only transformed. Furthermore, nature does not allow overdrafts. For example, you cannot burn tomorrow’s delivery of oil today, even if you have already paid for it. So, every fuel must have been energised by a greater source in the past, or somehow be re-energised from time to time.
Different forms of energy can be traded for one another, as happens in a working engine or generator. In any change energy tends to degrade and become less useful if left on its own. That is, boulders roll downhill but not up, and cups of coffee get colder but not hotter. To be practical, controllable and safe, an energy source must be stabilised somehow.
These criteria don’t leave many possibilities. For instance, most easily dislodged boulders rolled down hill long ago and most sources of heat have already cooled. All options available on a significant scale appear somewhere in the columns of the Table below.
| “Renewables” | Chemical (electronic) | Nuclear | |
|---|---|---|---|
| Fuels | Water, wind, sun | Fossil fuels,food, biofuels | Uranium, Thorium |
| Primed or renewed | Daily and seasonal sunshine | Sunshine in geological epochs | Pre-solar stellar collapse (supernova) |
| Energy density kWh/kg | 0.0003 | 1 to 7 | 20 million |
| Fuel for a whole life | 10 million tonnes | 1000 tonnes | 0.001 tonnes (1 kg) |
| Pro | Familiar, accepted | Reliable, available 24/7 | Reliable, safe, compact, resilient, available 24/7 |
| Con | Unreliable, weak, damaging to nature | Emissions, safety | Public apprehension, failed education |
Table of energy groups: the entries are explained in this essay
Three groups of natural energy sources
Possible sourcess fall into one of three distinct groups described by the columns of the Table.
The first column comprises the familiar energy sources of water, wind and sun. Everybody has confidence in these because no science is needed to appreciate their energy. Their mechanisms of falling, moving and warming are evident to the senses. However, the description “renewables” is not well chosen – they often fail to be renewed. Sunshine varies with the seasons and fluctuates unpredictably at the whim of the weather. The energy of tides and waves is similarly self-evident and also belongs in this group.
The second energy group includes all forms of combustion, including digestion. The only fuels like this naturally available on Earth on a grand scale are the fossil fuels – coal, oil and gas. Their energy is more enigmatic than “renewables”. There is no self-evident sign – no machinery, warmth or movement to suggest the hidden energy. Calling it “chemical” gives it a name, but does not explain what is happening.
There are other possible chemical fuels lack a natural production mechanism and a means to stabilise them. Even fossil fuels and wood are notoriously unstable in air and can be dangerous – the release of their energy is triggered by high temperature and so can spread. If it were possible on Earth to find large deposits of hydrogen, ammonia, or pre-charged lithium batteries, for instance, these would be candidates for primary fuels. They are certainly useful as secondary fuels, made or charged up using a primary source – but it is the choice of primary source that we are discussing here.
Technology is different from natural science. Natural science can tell us where there is energy. Then technology can be developed to find the most effective way to deliver it. But when nature says there is no energy, no amount of technical ingenuity or financial investment will find it. You cannot buy natural science.
The last column of the Table is the nuclear group. Like chemical, this is enigmatic, but also less familiar. It turns out that nuclear energy has an explanation similar to chemical energy.
Life and the supply of energy
Because energy is strictly conserved its supply must match demand, all the time and everywhere. So the electricity supplied by the grid has to be balanced, day and night, by the output and distribution from all the power stations, including the “Interconnectors” to other countries. On the internet you can watch how this happens with the different sources, country by country, all accounted in some detail and updated every few minutes.1 This is just part of a larger network of energy flows, including oil, coal and gas for transport and heating. If the energy runs out, many aspects of modern life stop – light, heat, transport, sewerage, internet, water.
The accounting of energy is ruthless and life has always struggled with it. Billions of years ago it made a great leap forward when it progressed from static plant life, largely dependent on short term sunshine, to mobile organisms, energised internally by stored fuel – that is food. Through evolution and selection over millions of years, animals, fish and birds learnt how to explore and source the food they needed and how to adapt to changing circumstances – those who did not learn to cope in a fluctuating environment died out. Evolution by natural selection is a tough school that leaves no room for the worthy features of humanity that society now prizes so highly today.
Energy revolutions that improved humanity
Humans have the advantage over other life forms of using their brain power to analyse their environment and rationalise their experiences. Consequently, they are able to adapt faster and avoid the punitive attrition characteristic of natural selection.
In the First Energy Revolution, starting some 600,000 years ago perhaps, humans realised ways to use energy beyond the digestive capacity of an individual. First, they used teams of animals and slaves; then the power of water and wind to drive mills and sail the world; also the heat of wood fires to cook food and to refine and fashion metals. Nevertheless, though better than animals, human beings at this time led short and miserable lives, and the population remained small. Ethical behaviour was for the few. Most struggled and suffered from disease and conflict, unremarked.
In the Scientific and Industrial Revolutions of the 17th to 19th Centuries however, mankind learnt to avoid sources of energy that depend on the capricious behaviour of wind and weather. By study and experiment society learnt how to engage the huge increase in energy available through coal and steam. With the new reliability life expectancy doubled and the population quadrupled. An emphasis could be placed on the virtues of humanity, including safety. With the high energy density of fossil fuels and their tendency to release energy contagiously or explosively, safety was particularly important. This was progressively improved by refining the design of machinery and by educating and training those concerned.
But now, twenty years into the 21st Century, the decision has been made to cease the burning of fossil fuel, so removing the very mainstay of the Industrial Revolution.
Every possible way forward is described in the Table. Its numbers and comments are crucial to future social and economic stability, living standards, impact on the environment and safety. But where do the entries in the Table come from, and what do they mean?
Energy density of visible sources
How much energy is available from “renewables”? The figure in the third row of the Table may be calculated using the mechanics of Galileo and Newton.
What does this mean for wind and solar, the most common “renewables”? Let’s calculate it.
So, solar and wind deliver similar amounts of power per square metre – rather modest amounts, in fact, but more can be harvested by simply building more square metres. But beware the impact on nature! The electricity demand in the UK is about 45 GW. If supplied by wind or solar that would require an area of several thousand square kilometres, and that would only work on a day when the sun was shining, and the wind was blowing.

There are equally unacceptable consequences when hydroelectric projects are scaled up. Recently major environmental problems are reported on the Nile, Mekong, Euphrates and Yangtse. These cause, not only damage to nature and massive displacements of people and homes, but enmity between whole countries. The coming water wars of the 21st Century are predictable.
Large “renewable” energy installations are easily damaged by freak storms and also incapacitated by changing weather patterns. They are easily sabotaged, too, by terrorists or hostile powers. In particular, off-shore wind farms are open to attack by enemy submarine.
Why can other types of energy source not be developed? The energy density of any “renewable” source that relies on movement is set by the speed alone. Tidal flow or waves may have an intuitive and attractive appeal, but the energy density is a similar number of kWh per kg, because the speed is similar. Material moving at the speed of sound has a higher energy density, about 0.01 kWh per kg – that is rather better. Molecules move at this speed in hot gases, but these are not widely available on Earth as a primary supply of energy.
In truth, any search for a new energy is easily led astray if it concentrates on energy that can be sensed, the ones we call classical. To choose the “touchy-feely” sources of energy may seem obvious, but it excludes the more energetic sources that nature has to offer. This is the mistake made by naïve environmentalism.
Invisible energy sources
Food that bought today is labelled with its energy density, printed on the packet. For example, “2018 kJ per 100g serving”, which translates as 5.6 kWh per kg, matching the fossil fuel values in the Table. These are far larger than the values for classical fuels. Why? What is the invisible energy mechanism? The quick answer is “Chemistry”, but a name is never an illuminating answer. What is the seat of this “super clockwork”?
For the electronic atom with the known electron mass m and the atomic size L, the Fermi Energy is about 4 electron-volts or 7 kWh per kg. The energy of all batteries, all chemistry, all lasers, all food, all explosives, all fossil fuels are on this scale and are explained as variations of this mechanism.
For protons and neutrons in a nucleus the same picture applies but the numbers are different. The nuclear size, L, is 100,000 times smaller than the atom and the proton mass, m, is 2000 times larger than the electron. The significant combination mL2 is five million times smaller, and the Fermi Energy five million times larger than for electron in the atom. This is the origin of the nuclear energy density shown in the Table. This “quantum clockwork mechanism” applies to both chemical and nuclear energies. However, the million-fold increase in energy density for nuclear changed the course of history after 1945. But, unfortunately, for seventy years nuclear energy was not appreciated by the people of the world as a benign source, a misunderstanding that is overdue for correction.

Core energy supply in the 21st Century
In use, electricity is the most flexible, clean and efficient form of energy, but it has to be generated from a primary source. As fossil fuels fall out of favour for heating and transport, the demand for electricity will grow further. However, the continuity of its supply is crucial, and a modern economy needs to be confident that it will be available 24/7. Which primary source of energy can be relied on to generate such a supply?
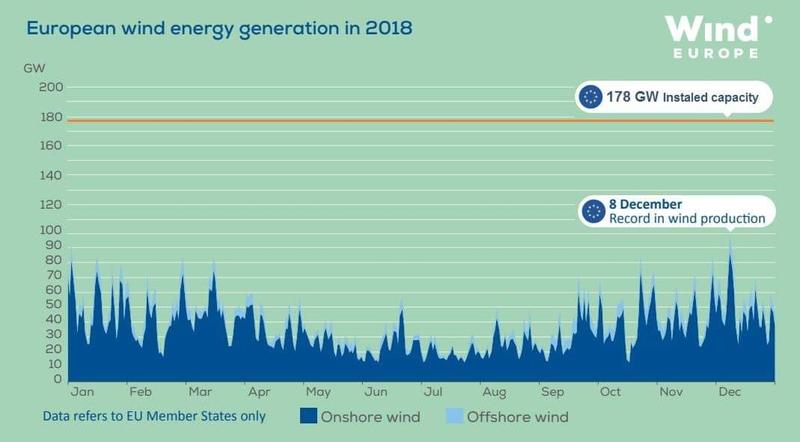
The strong point in favour of “renewables” is their popular acceptability. Against that are their unreliability, weakness and effect on the environment. Take the reliability of wind for example. In 2018 there was only one day on which the power delivered by wind reached 50% of its installed capacity in Europe as a whole (see the data published by Wind Europe). The average for the year was 22%, rising to 37% for offshore wind. This shortfall is worse than might be expected because the power depends on the cube of the wind speed (see the box above) – if the wind speed halves, the maximum available power falls to 12%. Given this intermittency, a significant back-up source is needed to avoid breaks in supply. Experience has shown that this is not possible without either nuclear or fossil fuels, and if the latter are excluded, that just leaves nuclear. Furthermore, since a nuclear plant can operate predictably at 90% capacity, any unpredictable contribution from “renewable” generation becomes superfluous to the maintenance of a reliable supply. If the power of a nuclear plant is reduced to balance a fall in demand or a drop in a parallel “renewable” supply, there is a small reduction in fuel cost but a related increase in the charge for idle capital. It is therefore more cost effective to dispense with “renewables” as a primary source and over-provide electricity from a nuclear plant running steadily at full power. The excess off-peak supply can be used to desalinate water and produce hydrogen or ammonia for transport, chemical feedstock and the domestic and industrial gas network. Unlike from a “renewable” plant, the large supply of waste heat from a nuclear reactor is also available for domestic and industrial heating. Such a complete unified power resource can be sited close to wherever it is needed, thereby reducing the requirement for expensive long-distance transmission and under-sea “interconnectors”.2
Electricity can be stored in batteries, but batteries are chemical, as first shown by Michael Faraday, and their energy density is correspondingly limited like fossil fuels. The only distinction is that batteries are “reversible”, meaning energy can be put into, or taken out of, a battery relatively easily by charging or discharging. Chemical battery technology has improved, but the idea that it might smooth the fluctuations of “renewables” over weeks and months will always be unrealistic. For example, a million tonnes of Tesla car batteries (150 GWh) could not supply the UK electric grid (45 GW) for more than an hour or two. Furthermore, the ability of batteries to deliver energy rapidly is intrinsically hazardous. It is likely that there will be serious battery accidents in the future and it is not clear that enough attention has been paid to this.

Most “renewable” energy comes originally from the Sun, and the energy of fossil fuels also came from the Sun but long ago in ancient geological time. But where does nuclear energy originate? The Thorium and Uranium mined today are the radioactive remnants of colossal stellar explosions powered by gravitational collapse that occurred before the Earth and Solar System were formed. Today astronomers study such supernova explosions elsewhere in the Universe. The picture shows a “modern” example, the remnant of the “Crab” Supernova, 6300 light years away, as seen today by the Hubble Space Telescope. The original explosion was visible for 23 days in broad daylight to Korean astronomers in July of the year 1054.
Nuclear safety
If the use of carbon fuels is to be reduced to zero in a few years, the implication for the deployment of nuclear energy is far reaching. The only mark against nuclear energy is public apprehension. Concern is widespread in society and rooted in the belief that nuclear energy is intrinsically unsafe.
Whatever the source, any large quantity of energy often excites fears for safety, especially if it is thought likely that control of it might be lost or fall into the wrong hands. Fossil fuels and chemical explosives are seen as providing many tales to support this. The huge extra power of nuclear energy only increases these fears. However, this overlooks an important distinction. Crucially, any release of chemical energy tends to drive positive feedback by increasing the temperature and spreading fire or detonating an explosion. So chemical combustion is intrinsically unstable, whereas the release of nuclear energy is not. A release can only be started if mediated by a flux of free neutrons. These neutrons do not exist in the wild because they decay in a few minutes and are rapidly absorbed in most materials anyway. So, energy generation by the neutron chain reaction is easily and completely shut down by absorbing the neutrons. In March 2011 all the reactors in Japan were shut down in this way as soon as the earthquake was detected. By the time the subsequent tsunami came ashore at the Fukushima Daiichi site, for instance, the energy being released was exclusively by radioactive decay. Radioactivity does not involve neutrons and cannot multiply. The activity released in the subsequent accident was not contagious and did not spread. Radioactivity is much safer than fire in this respect.
Radioactivity is not a disease. Someone who is contaminated or has been irradiated is not infected. Sadly, such people are shunned by others out of fear and ignorance following these rare radiological accidents. The wearing of chemical suits is seldom necessary for radioactivity, unlike for viral contamination. That radiation is not contagious should be a simple message of public health taught in schools. Why does that not happen? Unfortunately, there is a whole profession dedicated to explaining the dangers of radiation, and they do not allow themselves to question their dedication.
Arguably the most irrational safety fear concerns “the waste” produced by fission reactors. Because in quantity the fuel required is some million times less than the equivalent fossil fuel, the amount of “waste” is correspondingly tiny. Usually it is only a few percent “burnt” and so can be recycled, if required. The final fission waste is radioactive and needs to be cooled for a few years and then contained, as many chemical poisons need to be. But after 600 years, unlike a chemical, the radioactivity has decayed and is no more toxic than rock dug from the ground. Furthermore, waste cannot be used to make fuel for a nuclear weapon, as some people suppose. And then nobody has ever died in an accident from nuclear waste at a power plant. But, like other long-running media stories, “what to do with nuclear waste” still sells newspapers and refuses to die. It also provides jobs for many people and so – it is supposed – it must be important. Actually, the effort and the jobs are out of all proportion to any risk, and the work and its cost could be reduced dramatically.
Nuclear energy and its radiation have been used to benefit health ever since they were first discovered by Marie Curie and others. Uniquely, she was awarded two Nobel Prizes for her work in unravelling the physics and the chemistry of the science, and then dedicated her life to its application in medicine. Few members of the public, either personally or through friends and family, have not enjoyed longer and healthier lives as a result of diagnostic scans and therapy that use significant doses of radiation. Since radiation is undoubtedly powerful, the scientific question we need to answer is “why are moderate doses effectively harmless?”3
We all receive radiation naturally from rocks, space, the Sun and even radioactivity inside our own bodies. When life began on Earth about 3 billion years ago, these radiation levels were much higher than today. If biology had not evolved a series of effective protective strategies, we wouldn’t be here. This protection by evolution is remarkable, though it is only in the past few decades that some of the details have been understood. Evidence confirming that it works so effectively came from the accident at Fukushima Daiichi. Despite the anticipated disaster the significant release of radiation caused no injury at all and it was clear in a few days that none was likely.4
Marie Curie famously said, “Nothing is to be feared, it is to be understood”. But, if we do not understand, we are easily deceived. A benign example in medicine is the Placebo Effect. A diseased patient is told that he has been treated when he has not. Nevertheless, the message, despite the lack of treatment, increases his chance of recovery. The Nocebo Effect is the malign inverse – as in Voodoo. A healthy person is told that he has been cursed and will probably die. The likelihood is that he will begin to suffer with real symptoms, as well as significant mental health problems, unless of course he is able to expose this curse through understanding and confidence. Being told that you have been irradiated is such a curse. Even if you felt well previously, it is liable to damage your health. Surrendering to fear, individually or collectively, can lead to panic. The people at Chernobyl and Fukushima had no scientific understanding of radiation and their mental and social health were badly affected by the evacuation that followed. People around the world, too, were influenced by the combination of fear and excitement sent out by the media. Such excitement seen from the safety of an armchair in California or Germany has a potent effect on the world order despite calls to reason.4
But how can we be sure that radiation is harmless when the media supported by panels of “experts” suggest the opposite? The wildlife at Chernobyl was never told that it had been irradiated; it was not evacuated at short notice; it never watched a sensational video account of what happened; it never read a newspaper or registered for a compensation claim. Since it did not know and was not cursed, it received only the radiation. Did it suffer? How has the wildlife been affected since the human population left? Videos produced by the Discovery Channel and the BBC5 show animals thriving, better off being radioactive, but unencumbered by humans and free to roam undisturbed in a wildlife park. These suggest that radiation is more or less harmless, but the curse and Nocebo Effect are very serious. In similar experiments in the laboratory animals were divided into two identical groups, half were irradiated every day of their lives and half were not. These tests confirm that there is no effect for radiation levels several hundred times higher than recommended by the current precautionary regulations.6 Evidently the animals’ defences manage the effects of the radiation and no irreparable damage results, unless the radiation level is very high.
A comparison with the new virus, COVID-19, shows why nuclear is almost harmless. The virus has little energy but is dangerous because it is highly contagious and human life has no immediate protection against it. In time nature will evolve some immunity while the world’s biomedical laboratories are racing to develop a vaccine first. Gaining the required time and avoiding social and economic panic should be the priorities. Nuclear energy may be powerful but its effects are not contagious and immunity from it was a pre-requisite for life from the start three billion years ago. The most dangerous consequence of either an outbreak of a new viral infection or a nuclear accident is panic with its effect on social and economic stability.
Two conclusions: nuclear energy and education
Society was largely unaware of nuclear energy until the last days of the Second World War. First hearing about it in the context of war was an abrupt negative experience and not a helpful way to learn about an aspect of nature. In the decades that followed, talk of nuclear and radiation was shrouded in the secrecy and fear that were endemic in the Cold War. Concern about the escalating deployment of nuclear weapons led to highly disruptive political demonstrations. Attempts were made to maintain public order through appeasement. Outspoken public demand for protection against radiation led to recommendations approved by the United Nations. These were not based on firm evidence and were several hundred times more stringent than those internationally agreed in 1934. At the same time the perception that nuclear energy is unnatural, evil and beyond the understanding of most people discouraged its balanced study as regular part of natural science. This failure of general education has perpetuated radiophobia for seventy years.6
To select the best source of energy in the 21st Century society needs confidence. How may that be regained once lost? In the arena of fear and excitement confidence is a matter for daring and bravery, but in the digital world confidence is a matter for education, both for individuals and for society. For real confidence nobody should rely on authority, regulation or the word of assembled experts – they have their own tribal interests.
And it is not just about safety. Society needs to be sure its energy source is secure and available when needed. As Alice explained to the White Queen in Through the Looking Glass when offered jam, delivery yesterday or tomorrow is not good enough. Similarly, energy from Russia or the Middle East does not generate local confidence. Nor does a huge power plant serving a vast grid, a macho solution designed to impress fellow engineers and government officials. Confidence comes with a sense of ownership: a small plant blending unobtrusively into the landscape within twenty or thirty miles; a plant offering local employment and school visits; a plant of which locals can say “we generate our own electricity here, and send the surplus to the rest of the country”. The reality of the grid may be the same as a top-down approach, but the social perception of devolution is not. The coming variety of small nuclear reactors, factory built in modules and deployed locally, combined with scaled-down regulations based on science should reduce costs and timescales and provide power safely, acceptably and more cheaply than traditional sources.
The critical raw materials for the widespread adoption and scale-up of nuclear energy are know-how and public acceptance. Both depend on massive changes to education and culture. The biggest investment has to be made by those young people who enter the nuclear engineering field, ambitious to solve the technical problems and deploy the solutions. Their education takes time, but so too does the switch in general public perception needed to make nuclear and radiation matters acceptable. In time it can be done, just as over time smoking has become unacceptable. The gathering pressure of climate change and repeated supply failures will concentrate minds and encourage policy changes that should follow what is scientifically inevitable if civilisation as we know it is to survive.
Footnotes
-
The UK regularly imports 2 GW from France and 1 GW each from Belgium and the Netherlands. ↩
-
Video on Chernobyl wildlife (2012) Discovery Channel https://youtu.be/IEmms6vn-p8 and triggered pictures of wildlife at Chernobyl (2015) http://www.bbc.co.uk/news/science-environment-32452085 ↩
-
A much fuller discussion with references is given in the two books by Wade Allison Radiation and Reason (2009), and Nuclear is for Life (2015), available on Amazon and direct from the distributors at https://www.ypdbooks.com/science-and-technology/1690-wade-allison-special-book-pack-YPD01882.html ↩ ↩2
